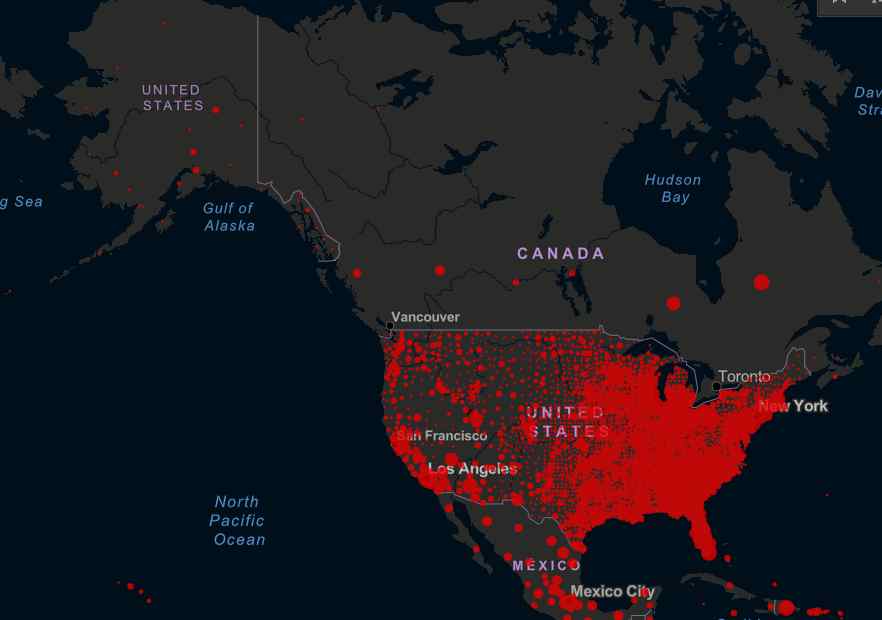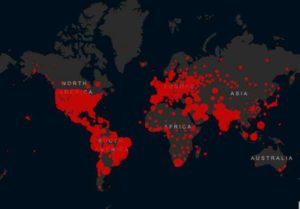
With the global death toll from COVID-19 pandemic now over 900,000, the top infectious disease expert in the United States says that drugmaker AstraZeneca’s suspension of final global trials of its experimental vaccine points to the effectiveness of the safeguards that have been incorporated into the trials.
“It’s important to point out that that’s the reason why you have various phases of trials, to determine if, in fact, these candidates are safe,” Dr. Anthony Fauci, the head of the National Institute of Allergy and Infectious Diseases, said in an interview with CBS This Morning Wednesday.
The British-Swedish pharmaceutical giant paused the trials because a volunteer participant became ill after receiving the experimental drug.
“It’s unfortunate that it happened, and hopefully they’ll be able to proceed along with the remainder of the trial,” Fauci said. “But you don’t know. They need to investigate it further.”
The company issued a statement Tuesday saying the pause in testing is a “routine action, which has to happen whenever there is a potentially unexplained illness in one of the trials, while it is investigated, ensuring we maintain the integrity of the trials.”
AstraZeneca developed the vaccine, AZD1222, in cooperation with Britain’s University of Oxford. The vaccine is being tested in large-scale Phase 2 and Phase 3 trials in several nations, including the United States, Britain, Brazil, South Africa and India. AZD1222 is one of three COVID-19 vaccines in late-stage Phase 3 trials in the United States.
[content id=”79272″]
The company did not disclose the nature of the participant’s illness, but The New York Times reports the volunteer, based in Britain, was diagnosed with transverse myelitis, an inflammatory syndrome that affects the spinal cord and is often sparked by viral infections. But the Times said it is unknown whether it is directly linked to the AZD1222 vaccine.
This is the second time AstraZeneca has paused large-scale testing of its experimental vaccine after a volunteer became ill after being inoculated. The scientific journal Nature says the trial was halted in July after another participant in Britain also developed symptoms of transverse myelitis. The individual was diagnosed with an “unrelated neurological illness.”
AstraZeneca is one of nine pharmaceutical giants pledging not to seek approval from U.S. government regulators for any vaccine until all data showed it was safe and effective.
The nine companies, including Johnson & Johnson, Merck, Moderna and Novavax, as well as those heading two joint vaccine projects, Pfizer and BioNTech, and Sanofi and GlaxoSmithKline, made the pledge in a statement released Tuesday, hours before AstraZeneca announced it was pausing its widespread testing of its vaccine.
The unusual joint pledge was aimed at alleviating growing fears by health experts that pharmaceutical companies are under considerable political pressure to quickly develop and introduce a COVID-19 vaccine. U.S. President Donald Trump has repeatedly suggested that a successful vaccine could be ready before November 3, the date of the U.S. presidential election.
British Prime Minister Boris Johnson announced Wednesday his government will launch an ambitious program to test at least half a million people daily for COVID-19, with the results back within minutes. The prime minister said he hoped the “moonshot” program — a reference to the 1960s-era American manned lunar program — will be in place before the end of the year, and would return Britain to some sort of normality and grant more freedom to those who test negative for the coronavirus.
Johnson coupled the announcement of the mass testing initiative with a new order limiting the number of people taking part in most social gatherings to six, from the current 30.
The new limit would take effect next week, as Britain is enduring a surge of nearly 3,000 new daily COVID-19 cases in recent weeks, the highest daily figure since May.
Britain’s Chief Medical Officer Chris Whittey says the new “rule of six” restrictions are likely to remain in place for the next several months.
Source: VOA